The focus on antibiotic-free poultry and livestock production means that everyone in the animal agriculture industry is inspecting, in detail, every aspect of the value chain. Similarly, as methods become more advanced, previously ignored factors that affect performance come to the foreground. One of these areas is the microbiome — in particular, the gut microflora.
More is being discovered about the relationship between an animal, its immune system and the microbiome. And with these developments comes the investigation of how they can be influenced. Feed ingredient and additive selection can greatly influence intestinal commensal bacteria and their interaction with other systems. Probiotics introduce beneficial bacteria to the gut, while areas under investigation are enzymes and peptidoglycans.
The microbiome
Just as in agriculture, medical science is changing its opinion of the microbiome and increasing understanding of its involvement in all aspects of human health. A paper published last year by three influential scientists stated, “The fitness of the host depends on, and cannot be seen as separate from, its microbiota.” It describes the interaction between the microbiome, the immune system, gut integrity and even oxidative status.
In livestock production, managing gut health is key to optimizing performance. And as the relationship between commensal bacteria and other physiological processes is more fully understood – the potential for manipulation increases. If there is an imbalance in the gut microflora, inflammation will occur, negatively affecting gut integrity. Not only does it make the animal more susceptible to infection, it may reduce nutrient absorption and lead to digestive upset.
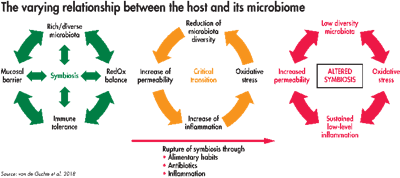
This diagram demonstrates three states of the microbiome: healthy gut (green), a critical transition of deteriorated symbiosis (orange) and a disease state (red). (van de Guchte et al. 2018)
Health and performance
Early gut development is important for all species, from birth or hatch. A healthy intestine will protect from infection and optimize nutrient absorption. It is these changes that mean better production and efficiency of livestock throughout their life cycle. Maintaining this state is also critical at weaning, or other diet change, when the gut microflora is susceptible to imbalance. Again, improving performance at this point sets the animal up to maximize its genetic potential.
How all the areas influenced by the microbiome work together to support health was described in the previously mentioned recent paper. A stable state, where health is maintained, is shown in green. In orange, there is a critical transition due to stress or challenge where symbiosis deteriorates, making the host more susceptible to disease. A disease state, in red, is where symbiosis has been altered.
Nutritional solutions
Enzymes are predominately used to improve the nutritional value of feed. However, their effect on the gut microflora can also influence performance. Research suggests that some carbohydrases have a prebiotic effect. By degrading non-starch polysaccharides, enzymes release oligosaccharides and some of these can be selectively used by beneficial bacteria in the gut, while others increase the production of short-chain fatty acids, particularly butyrate, by the gut microflora. This, in turn, improves immune activity within the intestine as well as its integrity. Results with poultry fed a synergistic combination of enzymes showed that changes in microbial populations improved performance.
The integrity of the mucosal barrier in the gut is essential for optimal health and performance. Here, nutrient absorption takes place and mucous is produced – preventing bacteria and toxins entering the blood stream. The gut, the most important immune organ in the body, works with the microbiota to prevent the need for costly inflammatory responses.
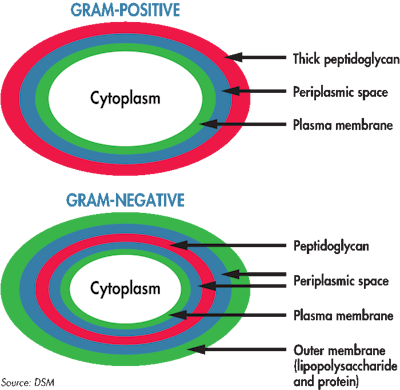
Gram-positive bacteria have a thick layer of peptidoglycan (PGN), shown in red, outside of the membrane. Gram-negative bacteria have significantly less PGN inside the membranes. (Courtesy DSM)
A look at peptidoglycans
Bacterial cell walls are composed of several layers: membranes, lipopolysaccharides, proteins and peptidoglycans (PGN). Gram-positive bacteria have a thick outer layer of PGN, while Gram-negative bacteria have a thin layer inside the membrane. PGN is a large amino acids/sugar polymer, which offers bacteria structural support against osmotic pressure. The molecule is continually growing and dividing, similar to process of bone metabolism. This high level of turnover means there is a large amount of PGN left in the intestine, particularly in livestock species, where Gram-positive bacteria predominate. While most of the intestinal flora don’t cause disease, once they die, they too contribute to intestinal waste. Animals are also exposed to PGN from feces, which attaches to dust particles and is breathed in from their housing environment.
PGN stimulates mucin secretion, which prevents pathogens and toxins entering the blood stream. High levels of threonine are required to produce mucin in the gut. In the case of increased stimulation, less threonine is available to the animal for growth, reducing performance and efficiency.
The innate immune system is able to recognize PGN as a microbial product. It then mounts an inflammatory response to a potential infection. This process is energetically expensive to the animal – again taking nutrients that would otherwise be used for growth. The inflammation itself means that nutrient uptake from the intestine is less effective and hence production efficiency is affected.
However, if the structure of the PGN can be altered, it may not be recognized by the host's defense systems, and animal performance would not be compromised by the mechanisms described above. The use of novel products to degrade PGN are being investigated as solutions to improve livestock production efficiency, again enzyme based. Most importantly, this technology doesn’t negatively affect the beneficial bacteria present in the gut – unlike antibiotics.
Probiotics for improved microflora
The effect of better microbial diversity in young broiler chicks will help gut development, improving future performance. For this reason, the practice of seeding the gut of chicks has been used for many years. Microbes are either sprayed onto the birds, dosed into the water, added to the feed or provided as a specific supplement. The options for probiotics products, for poultry and other livestock species, are numerous: Lactobacillus, Bacilli, Bifidobacteria, Enterococci and yeasts; single- or multi-species and combined with prebiotics or other additives.
By improving the balance of the gut microflora, the aim is to create an optimum environment to reduce the growth of pathogens, improve immunity and prevent inflammation – as well as enhancing digestion and nutrient absorption. In order to benefit performance, acting in all of these areas is important for consistent responses.
Stress relief
Much is talked about the effect of stress on animal performance: disease, environment, housing, nutrition, interactions, transport, etc. But there are other unobserved stresses on an animal’s physiology that divert nutrients away from growth and the immune system – hence production and efficiency. A stress on the animal’s microbiome is a stress on the animal itself. Gut health status is a result of interactions between intestinal integrity and a balanced gut microflora. Therefore, feed additives that can support these areas will benefit the health and performance of livestock.
In terms of positively influencing the microbiome, research is extensive, and solutions varied. This article has discussed the use of probiotics and enzymes. However, scientists are learning more and more about the relationship between the host and its microbiome. It is highly likely that new types of additive will be developed as scientific knowledge of this area grows.
References available on request.














