Given the ever-increasing rise in bacterial strains that are less and less sensitive to existing treatments, antibiotic resistance has the potential to become one of the greatest problems of our generation. While abuse of antibiotics in humans is probably the major contributor, policy makers have turned the spotlight on agricultural use as a way to control the problem.
Treatment of animals with antimicrobials can cause drug resistance to zoonotic pathogens. Bacteria from animals can be spread to humans via food products during slaughter and processing. This spread has been extensively documented for conventional foodborne pathogens, such as Salmonella, Campylobacter and E. coli.
Tracking antibiotic resistance zoonotic pathogens
Reliable data on antibiotic consumption and resistance is now quite easy to obtain. The European Union, for example, has implemented a coordinated approach to compiling relevant data since 2004. Within the EU, Sweden has had a monitoring program since the 1980s, and Denmark, via the DANMAP program, has provided an efficient record of antimicrobial consumption in humans and animals. In general, the more antibiotics are consumed, the more antibiotic resistance is found, regardless of whether such use is for therapeutic or growth promotant use.
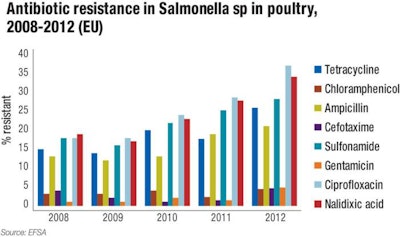
Based on the report “The European Union Summary Report on Antimicrobial Resistance in Zoonotic and Indicator Bacteria from Humans, Animals and Food,” Figure 1 highlights antibiotic resistance in Salmonella isolates from EU poultry.
European Food Safety Authority's (EFSA) review of the annual surveys on antimicrobial resistance, “The European Union Summary Report on Antimicrobial Resistance in Zoonotic and Indicator Bacteria from Humans, Animals and Food,” each year reveals some interesting, longer-term trends. Most notable is an increase in antibiotic resistance among Salmonella isolates in poultry (Figure 1). In particular, and of concern for human health, there has been an increase in the resistance for Salmonella enteritidis the past few years (Figure 2).
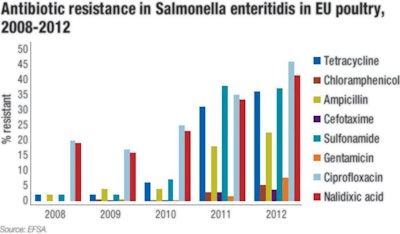
Figure 2 charts antibiotic resistance in Salmonella enteritidis found in EU poultry from 2008 to 2012.
Other trends indicate that the relative resistance rates for S. typhimurium remain static, but this should still be a concern due to the relatively high rates noted.
The majority of antibiotic resistance can be noted among broilers, and this may in part be due to the lower level of antibiotic use in the layer industry relative to the broiler market. The data also indicates a trend towards an increase in antibiotic resistance in Salmonella isolated from poultry meat since 2010. These temporal changes would need to be monitored for a longer period to determine the extent of this rise; however, there is a relatively high rate among some classes of antibiotics, e.g., nalidixic acid, ciprofloxacin and the sulphonamides.
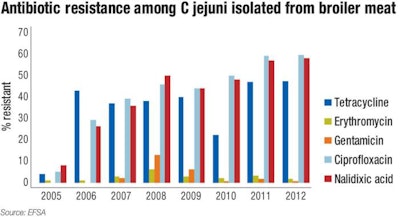
Figure 3 shows the antibiotic resistance patterns of C jejuni found in broiler meat.
The data further indicates a high level of antibiotic resistance for Campylobacter sp., with temporal trends indicating a rise in resistance among specific strains and to specific antibiotics. Of particular interest is the rise in resistance to antibiotics, such as nalidixic acid and ciprofloxacin in C jejuni and C coli in broiler meat. High level resistance to these antibiotics has been noted in the same strains when isolated from humans.
Curbing antibiotic usage with additives
While the debate rages over what is driving the seemingly inexorable rise of antibiotic-resistant microbes, alternatives to antibiotics and products capable of reducing the risk of antibiotic resistance transfer through the food chain are clearly required.
Globally, it is recognized that there is no so-called “silver bullet” to replace antibiotic use in animal production and producers will almost certainly have to improve hygiene and husbandry to address the issue.
Products that will assist the move to antibiotic-free production status include many that are designed to regulate and support the gut environment and its microflora:
- Coccidial vaccines
- Probiotics
- Competitive exclusion products
- Feed enzymes
- Functional nutrients such as nucleotides
- Organic acids and feed hygiene products
- Organic minerals
- Plant-based products such as herbs, spices and essential oils
- Yeast cell wall derivatives such as mannan-oligosaccharides (MOS) and mannose rich fractions (MRF)
Of the functional ingredients currently in use for microbial control, MOS and MRF are widely used in animal nutrition and have been shown to improve animal performance in a manner similar to antibiotic-like growth promoters.
Exploring yeast cell wall derivatives
Since 1999 the use of MOS in animal feed has become more prominent, mainly due to the European ban on prophylactic antibiotic growth promoters in animal feed. Given their ability to bind and limit the colonization of gut pathogens, MOS and MRF have proven to be an effective solution for antibiotic-free diets, as well as providing support for immunity and digestion.
Some newer studies have focused on the effect of MRF supplementation on the overall bacterial community of the poultry gut and such work has shown that MRF supplementation can significantly alter the intestinal microflora or the so-called microbiome.
More strikingly, consistent alterations in specific components of the caecal microbiota of broilers have been identified with alterations also noted in numerous biological pathways as a result of supplementation with MRF.
One strategy to reduce or limit the use of antibiotics is to find ways to make them more effective. The use of MRF has been shown to enhance the sensitivity of bacteria to the effects of antibiotics and in doing so, reduce the minimum inhibitory concentration (MIC) required. Additionally, MRF supplementation to the diets of broilers has been associated with a decrease in selected antibiotic resistance gene copy number. This is potentially linked to the ability of MRF to induce plasmid loss, thereby, reducing plasmid transfer between microbes (Table 1).
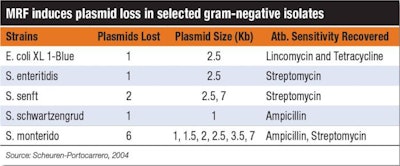
Scheuren-Portocarrero, 2004 | Table 1 demonstrates MRF’s ability to potentially reduce plasmid transfer between microbes.
It is essential that antibiotic-free production systems improve overall feed quality as animals that are fed quality feeds are less susceptible to enteric problems. Ultimately this move from least-cost feed formulation and reliance on antibiotics will be toward high-quality feeds containing functional ingredients.
Programs that employ a holistic approach to health management have proven to be extremely effective. By utilizing a combination of strategies, producers can rehabilitate and accelerate the evolution of the intestinal microbiota. The success of these “Seed, Feed and Weed” programs is reliant on firstly seeding the gut with favorablemicroflora using a probiotic; feeding the favorablemicrobes through acids or enzymes; and, lastly, weeding out pathogens by using MRF products.

















