Mineral utilization by animals depends primarily on their absorption from the ingested feed, with the term “bioavailability” generally being used to describe both the absorption and the ultimate metabolic utilization of nutrients. In the past decade, the feed industry has experienced a fine-tuning in diet formulation, not only to match the animal’s nutritional needs, but also to minimize pollution due to mineral excretion.
There are many forms of mineral complexes and chelates available for use in animal nutrition; these have (perhaps unfortunately) been generically titled organic trace minerals (OTM) by virtue of the fact that the minerals in question are bonded to organic molecules such as amino acids or peptides, for instance.
The bioavailability of OTMs
An important feature of OTMs is their enhanced stability due to the conformation in which the metal is held by the bonding group(s).
In general terms, the stability of an OTM is the defining characteristic of its bioavailability. During transit through the GI tract, all OTMs are subjected to changes in pH that can result in the bound mineral complex dissociating and releasing free mineral ions. As a result, the charged free mineral ion can react with plant components such as phytic acid or can undergo hydroxy-polymerization resulting in precipitation of the mineral. These can both lead to a very significant reduction in mineral bioavailability.
Additionally, in the case of copper and iron (Cu2+ and Fe2+), this can cause oxidative stress via the production of free radicals, and as such increase the requirement for vitamin E as well as for selenium and vitamin C, which are required for vitamin E recycling.
Essentially, OTMs with low stabilities reduce the effectiveness of the product to that of the corresponding inorganic salt.
Negative interactions
The possibility for negative interactions occurring between individual components within premixes and feeds is high and often overlooked. For instance, exogenous sources of phytase and trace elements are routinely supplemented to monogastric diets and usually combined in a premix. Even though metal ions can act as enzyme inhibitors, little thought is given to the potential for mineral-induced inhibition of enzyme activity.
A recent peer-reviewed study focused on assessing the potential in vitro interaction between inorganic and organic sources of Fe, zinc (Zn) and Cu with three commercially available phytase preparations. The study also investigated if the degree of enzyme inhibition was dependent on the type of OTM used as mineral source.
The authors demonstrated that a highly significant relationship between phytase inhibition, trace mineral type as well as mineral source and concentration existed.
Of the mineral sources tested, proteinates were consistently and significantly less inhibitory than the majority of the other sources.
IC50 values for phytase inhibition by the individual mineral sources
The half maximal inhibitory concentration (IC50) values represent the concentration of minerals at which the phytase activity has been reduced by 50 percent and allows the reader to quickly determine the relative extent of enzyme inhibition. (See Tables 1-3)
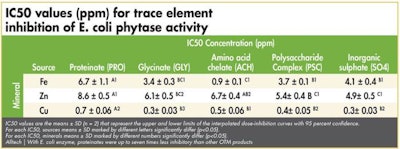
Alltech | With E. coli enzyme, proteinates were up to seven times less inhibitory than other OTM products.
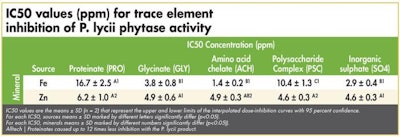
Alltech | Proteinates caused up to 12 times less inhibition with the P. lycii product.
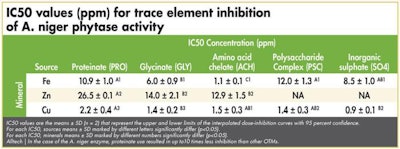
Alltech | In the case of the A. niger enzyme, proteinate use resulted in up to 10 times less inhibition than other OTMs.
In general terms, the authors concluded that, in the case of the E. coli enzyme, proteinates were up to seven times less inhibitory than other OTM products. They showed that, for the P. lycii product, proteinates caused up to 12 times less inhibition and in the case of the A. niger enzyme, proteinate use resulted in up to10 times less inhibition than other OTMs. Significantly, the authors demonstrated that, in the case of some OTMs, the inhibition they caused was on par with that caused by the inorganic variant.
Overall, different OTM sources displayed differential effects in their inhibition of exogenous phytase activity. This differential effect indicates that not all OTMs are created equal; moreover they all differ in terms of their stabilities. This indicates that OTMs release minerals in a pH-dependent fashion based on the pH in the local micro-environment.
The consequences that this mineral induced inhibition of enzyme activity has for premix and feed formulation are tremendous. It may well be that the trend toward super-dosing of phytase activities in diets is an unintentional consequence of the negative interactions of premix components. Ultimately, diet formulators may need to pay greater attention to their choice of individual component.
Conclusions at a glance
- Different OTM sources have differing abilities to inhibit exogenous enzymes such as phytase
- Proteinates were the most stable source of mineral and caused least inhibition of phytase activity
- Inorganic minerals are highly inhibitive of phytase
- The findings of this study are a potential indication of the interactions that can occur within a premix and care should be taken when choosing individual components.














