Organic acids have been used extensively since the time growth-promoting antibiotics were banned in the EU. They constitute one of the most widely accepted alternatives to traditional antimicrobial agents, but their effectiveness depends on their potency, concentration, synergistic effects with other organic acids and other additives, and stomach pH that fluctuates with age.
Despite their widespread use and very extensive technical and research literature on the subject, there is still a considerable gap in our knowledge that goes beyond the classic dose-response effect as these compounds require knowledge of basic chemistry before using them correctly.
What are these organic acids?
Organic acids exist in most vegetable matter we consume every day. For example, citric acid, a common organic acid, is present in most fruits and in great quantities in citrus fruits. Malic acid is found in apples, whereas the common vinegar is acidic acid. By nature, organic acids are rather weak, as evidenced by their widespread presence and safe consumption by humans and animals. On the other hand, they can be potent enough antimicrobial agents when in sufficient concentration, but like any such compound, the required dosage depends on the pathogenic load. That is, the more bacteria present, the higher the dosage is required to neutralize them.
How do they differ from inorganic acids?
Inorganic acids are equally widespread in nature, but they are found in very minute amounts unless industrially concentrated. For example, two common inorganic acids include phosphoric acid and hydrochloric acid. The first is used in many soda drinks, whereas the second is secreted by the stomach of animals. Inorganic acids are strong acids in that they can cause damage if not diluted greatly. For example, highly diluted hydrochloric acid can be handled with safety by our stomach, but concentrated will just burn through it. It is evident that the use of inorganic acids poses significant safety problems in the whole manufacturing process. Thus, although they are quite effective as disinfectants (such as the commercial chlorine solutions), they are not commonly used as feed additives.
How do we define the strength of organic acids?
We have referred to organic versus inorganic acids as weak versus strong, but this is a qualitative term that needs to be backed up by a numerical index that can be used to compare acids on an equal footing. The term that has been agreed upon by chemists worldwide is that of pKa, which is nothing but the negative logarithm of the concentration of reaction participant bodies at equilibrium. It is a rather complicated index when it comes to its definition and calculation. In biochemistry terms, and this is where we need this index to help us understand organic acids, we can understand pKa as the pH at which 50 percent of the acid is dissociated. In even more practical terms, the higher the pKa, the weaker our acid will be.
How organic acids benefit the animal
Organic acids are believed to affect pathogenic bacteria through two main ways. First, they can reduce the pH of the surrounding environment, making it impossible for undesirable bacteria to survive. This role is usually performed by stomach hydrocloric acid that kills most external bacteria consumed along with food or water, but it does not affect naturally present bacteria that just consume nutrients. For organic acids to lower stomach pH, aiding or substituting naturally secreted hydrochloric acid, we need strong(er) acids that split (dissociate) under stomach conditions.
Read more: Six questions concerning organic acids
Second, organic acids in their intact (un-dissociated) form are believed to penetrate the bacterial outer membrane entering its cytoplasm where they dissociate under a neutral pH. This drops internal pH, and bacteria eventually die while trying to overcome the problem. Only weak acids remain intact in the stomach, entering into the small intestine where they can attack bacteria. Such action also occurs in the stomach, but it is of limited significance because feed does not stay long in there. In contrast, in the small intestine, bacterial populations are substantial, and this is where a general reduction in good and bad bacteria usually ends up improving feed conversion efficiency. However, the action of organic acids can be curtailed by the overpowering ability of the organism to maintain neutral pH in the intestines, causing organic acids to dissociate sooner or later. To this end, only covered or otherwise protected organic acids can reach the large intestine.
What is the importance of pKa in the stomach?
We usually pick organic acids to act in the small intestine, ignoring the importance of having a low pH barrier in the stomach, but this requires some explanation. In the stomach of animals, there is water and hydrochloric acid, with the resulting pH being about 2, which is very acidic — as needed to bar entrance to most pathogens. In young animals, with a still developing digestive system, pH is often closer to 4, especially in animals that have been just weaned from an all-milk diet. It is important, then, to understand how organic acids fare in these two stomach pH values, and this will lead us to select the right acids for the right purpose.
For that, let us consider first lactic acid (a weak organic acid) with a pKa of 3.86. At stomach pH 2 (mature animals), the majority of acetic acid will remain intact (un-dissociated and thus ineffective in reducing stomach pH). In contrast, at a stomach pH of 4 (young animals), the majority of lactic acid will split to its components releasing hydrogen protons, which drop pH, and the conjugate base. Thus, in its dissociated form, lactic acid at pH4 will be effective in reducing stomach pH, at least until pH is reduced to below its pKa, but not suitable as an antibacterial agent in the small intestine since the majority of it will be consumed in the stomach.
Next, let us consider acetic acid (an even weaker organic acid) with a pKa value of 4.75. It is evident that this acid will remain largely intact, and thus ineffective in reducing stomach pH, in both young and mature animals as its pKa is higher than any of the expected stomach pH values. This is not important, of course, for mature animals, but it demonstrates that in young animals acetic acid is a rather poor choice to reduce stomach pH.
Finally, there is the case of organic acids with more than one pKa value, such as citric acid with three pKa values as it has three protons to give in solution: 3.13, 4.76, 6.40. It is evident that the first dissociation of protons will occur in the stomach of young animals, whereas the other two will have to wait until they reach the small intestine (where pH is held near 7). Of course, some of the citric acid will be absorbed by the stomach wall cells as a direct energy source, but this is a totally different story.
Mixes of organic acids
Here it must be noticed that so far we have been discussing the behavior of organic acids in the stomach — something that is largely ignored. In the small intestine, where pH nears 7 (neutral), organic acids continue to act indiscriminately against bacteria as dissociation becomes virtually complete. Therefore, the value of an organic acid should be determined by the location at which it is desired to act. Some organic acids are more suitable for the stomach (to prevent pathogens from entering the gut), whereas others are required to act at the intestines level (to reduce total bacterial load). To this end, it is common practice today to use mixes of organic acids that cover a broad pH spectrum for different locations in the gut. In addition, not all organic acids are equally effective against all pathogens, but this again requires a different line of discussion.
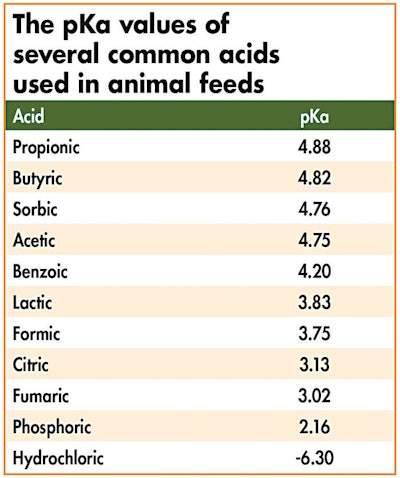
It is evident that hydrochloric acid is a most potent acid as it dissociates completely and instantly in water solutions.
Sources of pKa values
There is a plethora of tables available in chemistry books and on the internet presenting pKa values for most known organic acids. A comprehensive guide of over 1,000 compounds from Colorado State University can be found on their website.